Lama laba combination copd12/30/2023 
Indirect comparisons: Six NMAs were identified that reported indirect comparisons between LAMA/LABA FDCs five of these were within the pre-defined scope of this review. However, the differences in study methodology and patient characteristics between these studies made it difficult to draw generalizable conclusions regarding the comparative effectiveness of LAMA/LABA FDCs from the direct comparisons alone. 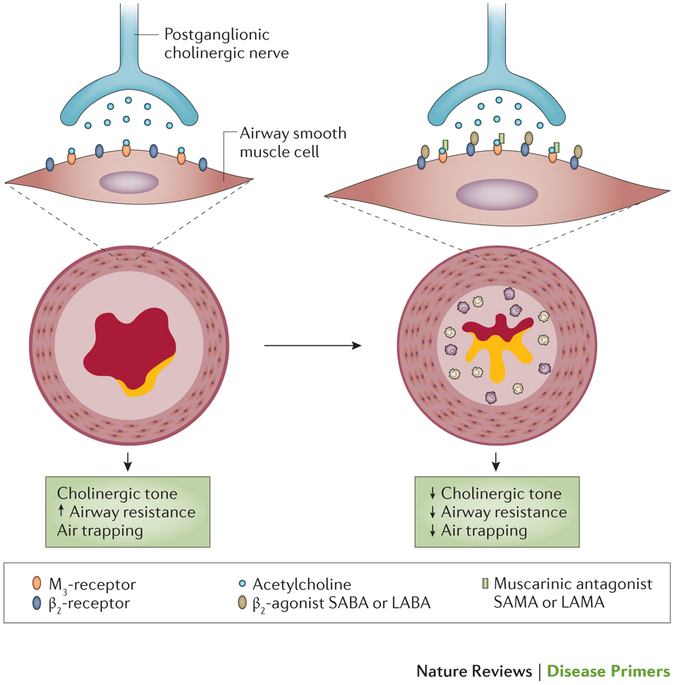
While some differences in lung function outcomes were noted, where assessed, LAMA/LABA FDCs had comparable efficacy in improving symptoms, health status, exacerbations, and comparable safety profiles. The review of indirect comparisons focused on NMAs reporting efficacy outcomes at 12 and 24 weeks of treatment (established durations of symptomatic studies in COPD recommended by regulators).ĭirect comparisons: Four RCTs that provided head-to-head comparisons of LAMA/LABA FDCs were identified, and these varied in their study design, included patient population and reported endpoints. Study/Analysis characteristics, eligibility criteria, patient characteristics, and overall conclusions were extracted from relevant publications. Two systematic literature reviews were conducted to identify direct comparisons (head-to-head randomized controlled trials ) and indirect comparisons (network meta-analyses indirect treatment comparisons meta-analyses) in patients with COPD with moderate-to-very severe airflow limitation. This literature review assessed comparative efficacy and safety of long-acting muscarinic antagonist/long-acting β 2-agonist (LAMA/LABA) fixed-dose combinations (FDCs) in patients with COPD and moderate-to-very severe airflow limitation, using evidence from direct (head-to-head) and indirect treatment comparisons.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
